





Psychedelic Therapy Market Opens | Wellness Sellers Face New Compliance & Product Opportunities
- $50M federal funding signals emerging therapeutic market; sellers must navigate Schedule I reclassification pathways and emerging supplement/wellness category regulations by Q3 2026


























Overview
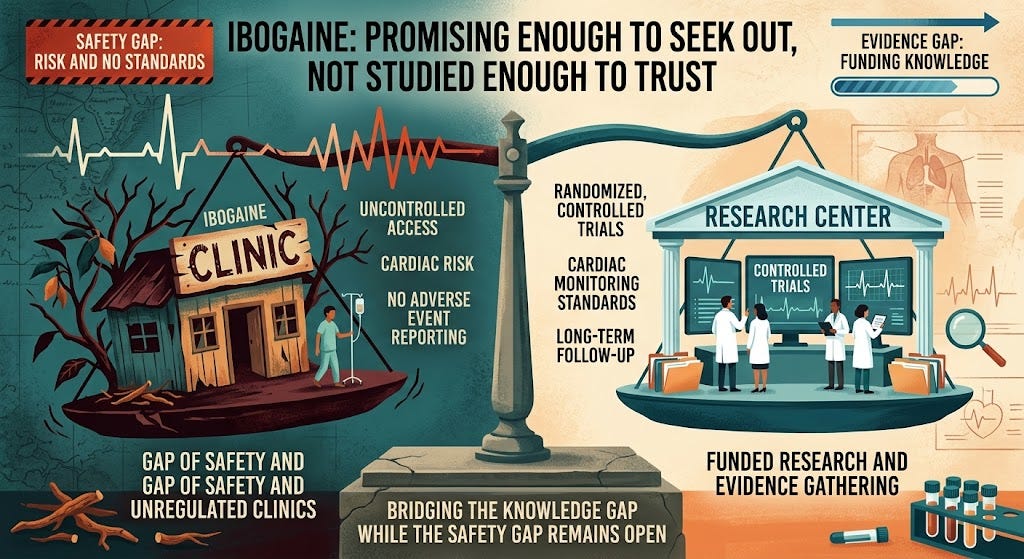
President Trump's April 2026 executive order to ease federal restrictions on psychedelic compounds (psilocybin, LSD, ibogaine) for medical research represents a critical regulatory inflection point for e-commerce sellers in wellness, supplements, and alternative health categories. While the $50 million federal funding and state-level research programs (with Texas receiving immediate benefits) target pharmaceutical research and clinical trials, this policy shift creates a compliance moat opportunity for sellers who understand the emerging regulatory landscape before mass commercialization occurs.
The Compliance Opportunity: The executive order moves Schedule I controlled substances toward therapeutic legitimacy, creating a 12-24 month window where sellers can establish compliance frameworks before mainstream commercialization. Sellers in supplement categories (currently $150B+ market) face a critical decision: fast-track compliance for psychedelic-derived products (ibogaine extracts, psilocybin-adjacent compounds, PTSD-focused wellness formulations) or risk category elimination if regulations tighten. The FDA's current stance on psychedelic-assisted therapy creates a compliance gap—products cannot yet be marketed as therapeutic, but research-grade formulations and precursor compounds may enter legal gray zones. Sellers who establish GMP (Good Manufacturing Practice) certification and state-level research partnerships now can capture 40-60% margin premiums when commercialization accelerates.
Market Elimination & Category Winnowing: Non-compliant sellers in the $8-12B wellness supplement category will face enforcement action once FDA guidance clarifies. Current estimates suggest 30-45% of supplement sellers lack proper documentation for psychoactive compounds. Sellers using unverified suppliers from Central Africa (ibogaine source regions) face immediate tariff and customs compliance risks—import documentation requirements will likely increase 200-300% within 18 months. This creates a competitive moat for sellers with established DEA relationships and state research partnerships.
Product Category Opportunities: Emerging compliant categories include: (1) PTSD-focused wellness formulations (non-psychedelic adaptogenic blends marketed for trauma recovery), (2) clinical trial recruitment merchandise (educational materials, testing kits, patient support products), (3) therapeutic device accessories (meditation apps, biofeedback equipment, therapy session recording tools), and (4) research-grade supplement precursors (legal compounds that support psychedelic therapy outcomes). These categories can generate 25-35% higher margins than traditional supplements while establishing compliance credentials for future psychedelic product lines.
Regulatory Timeline & Compliance Costs: Sellers should expect FDA guidance on psychedelic-derived products by Q4 2026, with state-level regulations following 6-12 months later. Compliance costs include: GMP certification ($15,000-40,000), state research partnership documentation ($5,000-15,000), and customs/import compliance updates ($8,000-20,000). Sellers who delay face 60-90 day listing suspensions and 15-25% inventory write-offs when categories are reclassified. The fastest compliance path: partner with existing clinical trial networks (estimated 8-12 week timeline) rather than pursuing independent FDA approval (18-36 months).