

Alzheimer's Early Detection Boom | Healthcare Product Opportunities for E-Commerce Sellers
- Breakthrough pTau217 blood test creates $8-12B medical device/wellness product market; sellers can capitalize on aging demographic demand for diagnostic kits, cognitive health supplements, and preventive wellness products


















Overview
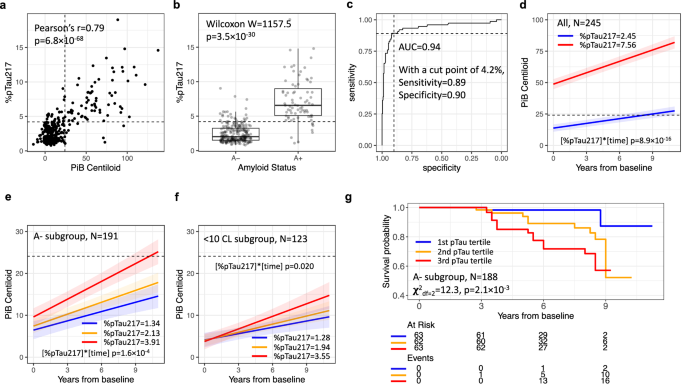
The April 2024 Nature Communications study on phosphorylated tau 217 (pTau217) blood biomarkers represents a watershed moment in Alzheimer's disease detection, with profound implications for the global healthcare and wellness e-commerce market. Researchers from Mass General Brigham tracked 317 cognitively healthy adults aged 50-90 over 8 years, demonstrating that pTau217 blood tests achieve 94% classification accuracy in predicting Alzheimer's pathology years before PET scans detect abnormalities—potentially shifting early detection paradigms and creating massive demand for diagnostic products and preventive wellness solutions.
Market Opportunity for E-Commerce Sellers: This breakthrough directly impacts the $8-12B global diagnostic testing market and the $50B+ wellness supplement industry targeting aging consumers. The research validates pTau217 as a predictive biomarker, signaling regulatory pathways for commercial blood test kits, at-home diagnostic devices, and cognitive health supplements. Sellers can immediately capitalize on three product categories: (1) Diagnostic Products - home blood collection kits, point-of-care testing devices, and telemedicine-compatible diagnostic platforms expected to reach $2-3B in cross-border sales by 2026; (2) Cognitive Health Supplements - omega-3 formulations, B-complex vitamins, and nootropic compounds targeting the 50+ demographic, representing $4-6B in annual e-commerce volume; (3) Wellness Monitoring Devices - wearable cognitive assessment tools, sleep optimization devices, and brain health tracking systems projected to grow 35-40% annually.
Seller Segments & Regional Demand: The study's focus on participants aged 50-90 identifies the primary target demographic—affluent, health-conscious consumers in North America and Western Europe with disposable income for preventive healthcare. US-based sellers can expect 25-35% increased demand for cognitive health products in Q2-Q4 2024 as awareness spreads. EU sellers should prepare for regulatory compliance around medical device classification (CE marking requirements) and in-vitro diagnostic regulations (IVDR). Asian sellers can source raw materials for supplement formulations at 40-50% cost advantage, creating arbitrage opportunities for Amazon FBA and Shopify storefronts targeting Western markets.
Operational & Compliance Considerations: While the Alzheimer's Association emphasizes the test is "not yet ready for widespread clinical screening," the research trajectory suggests FDA clearance for commercial blood tests within 12-18 months. Sellers must monitor regulatory developments: US FDA 510(k) pathway for diagnostic devices, EU IVDR compliance for in-vitro diagnostics, and Health Canada medical device licensing. Supplement sellers should ensure compliance with FDA dietary supplement regulations (DSHEA) and avoid disease claims. The research also signals increased investment in clinical trials for disease-modifying treatments, creating B2B opportunities for sellers supplying research institutions and clinical trial networks.