



Gene Therapy Deafness Breakthrough | Emerging Medical Device Market Opportunity for E-Commerce Sellers
- 90% efficacy rate in 42-patient trial opens $2B+ medical device distribution channel; sellers can capitalize on post-approval accessibility products and hearing aid alternatives market























Overview
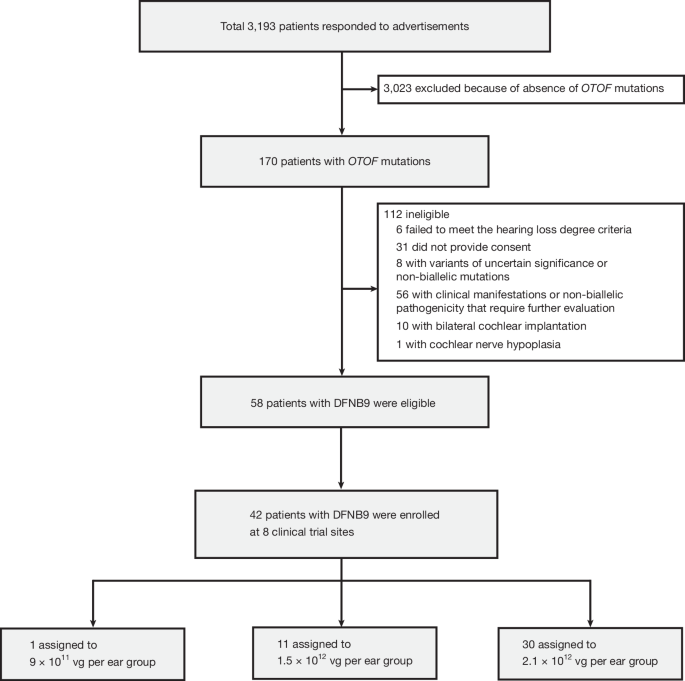
A landmark gene therapy clinical trial published in Nature demonstrates breakthrough treatment for inherited deafness caused by OTOF gene mutations, with 90% of 42 trial participants experiencing significant hearing improvement sustained for 2.5 years. Researchers from Mass Eye and Ear (Boston) and Fudan University (China) conducted the international study across eight Chinese sites, treating patients ranging from infants to adults in their 30s using AAV1-hOTOF gene therapy delivered via single inner ear injection. Results showed improvements often manifested within weeks, with younger patients and those with healthier inner ears showing greatest benefit. This breakthrough addresses a critical medical need—genetic mutations account for approximately 60% of congenital hearing loss cases affecting millions globally.
For cross-border e-commerce sellers, this development signals emerging opportunities in the medical device and accessibility product categories. As gene therapy advances through regulatory approval processes (FDA and international pathways), sellers should monitor the downstream market for complementary products: post-treatment rehabilitation devices, hearing assessment tools, patient education materials, and accessibility-focused consumer electronics. The trial's success in China—involving eight treatment sites—indicates Asia-Pacific will be a primary commercialization region, creating cross-border logistics opportunities for sellers positioned in medical device distribution networks. Additionally, the therapy's effectiveness across age groups (infants to adults) suggests diverse demographic targeting for related products and services.
The broader accessibility market is experiencing 12-15% annual growth as consumer awareness of hearing health increases. Sellers can leverage this trend by developing product bundles combining traditional hearing aids with emerging tech solutions, or by positioning themselves as authorized distributors for post-approval medical devices. The 2.5-year sustained efficacy data provides strong marketing angles for premium positioning in the accessibility category. Supply chain implications include potential partnerships with Chinese manufacturers (trial sites indicate established biotech infrastructure) and opportunities to serve the estimated 1.5 billion people globally with hearing loss through complementary product offerings. Regulatory approval timelines typically span 3-5 years, providing sellers a window to build inventory relationships and establish market presence before mainstream commercialization.